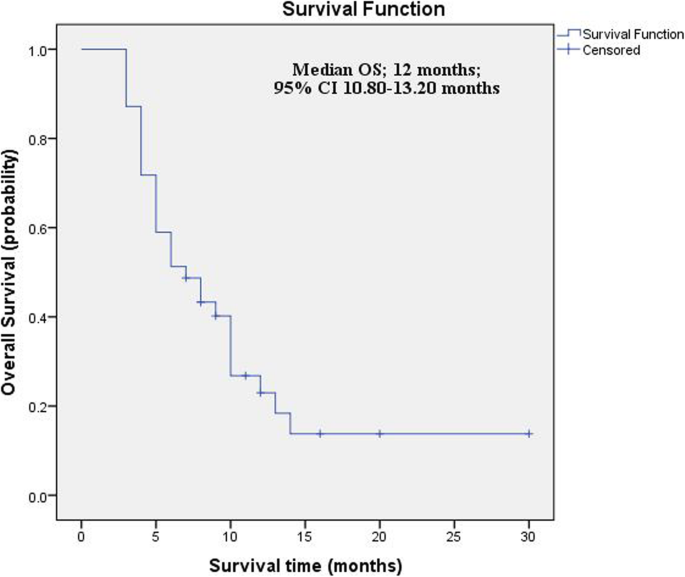
Overall survival (OS) analyses. Case numbers, median OS and P-values... | Download Scientific Diagram

Overall survival (OS) of all patients. The median OS is 26 months (95 %... | Download Scientific Diagram
Comparison of median OS (overall survival) and PFS (progression free... | Download Scientific Diagram
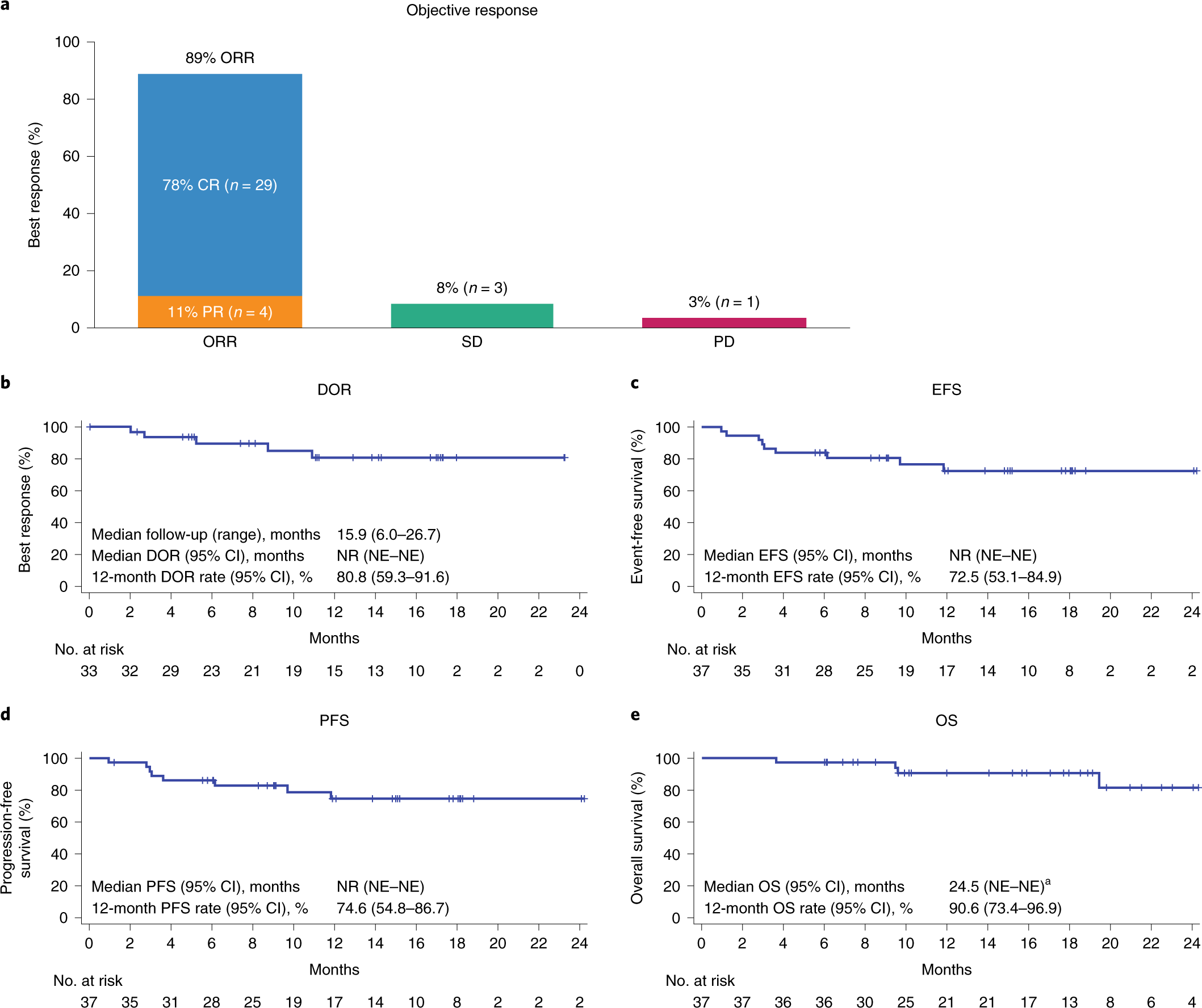
Axicabtagene ciloleucel as first-line therapy in high-risk large B-cell lymphoma: the phase 2 ZUMA-12 trial | Nature Medicine
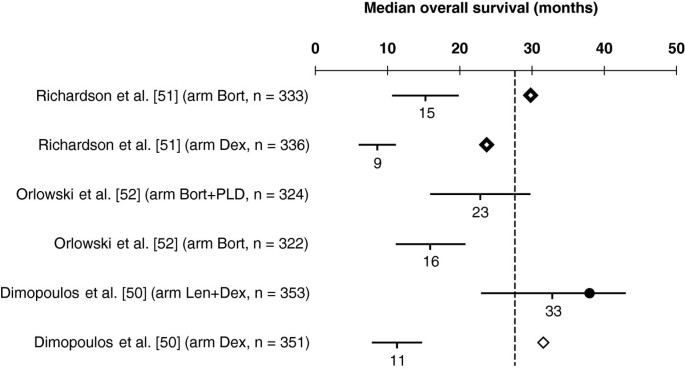
Time-dependent endpoints as predictors of overall survival in multiple myeloma | BMC Cancer | Full Text
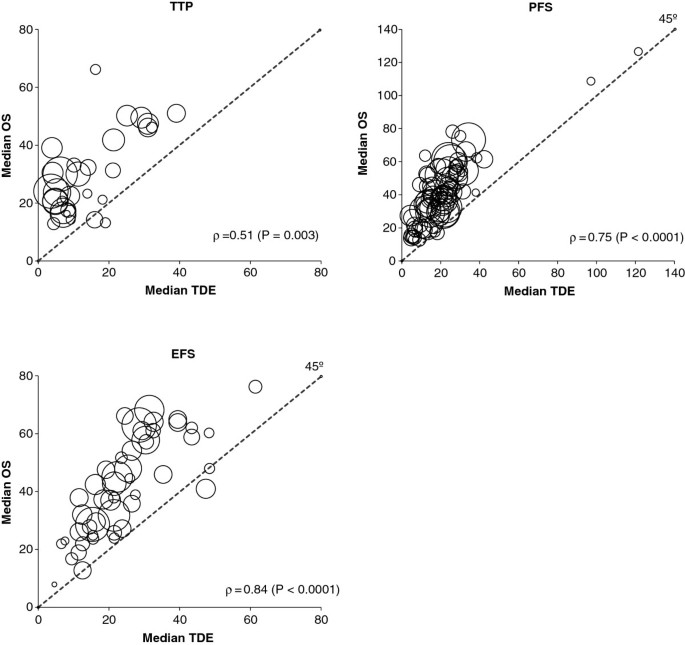
Time-dependent endpoints as predictors of overall survival in multiple myeloma | BMC Cancer | Full Text

Assessing the efficacy‐effectiveness gap for cancer therapies: A comparison of overall survival and toxicity between clinical trial and population‐based, real‐world data for contemporary parenteral cancer therapeutics - Phillips - 2020 - Cancer -
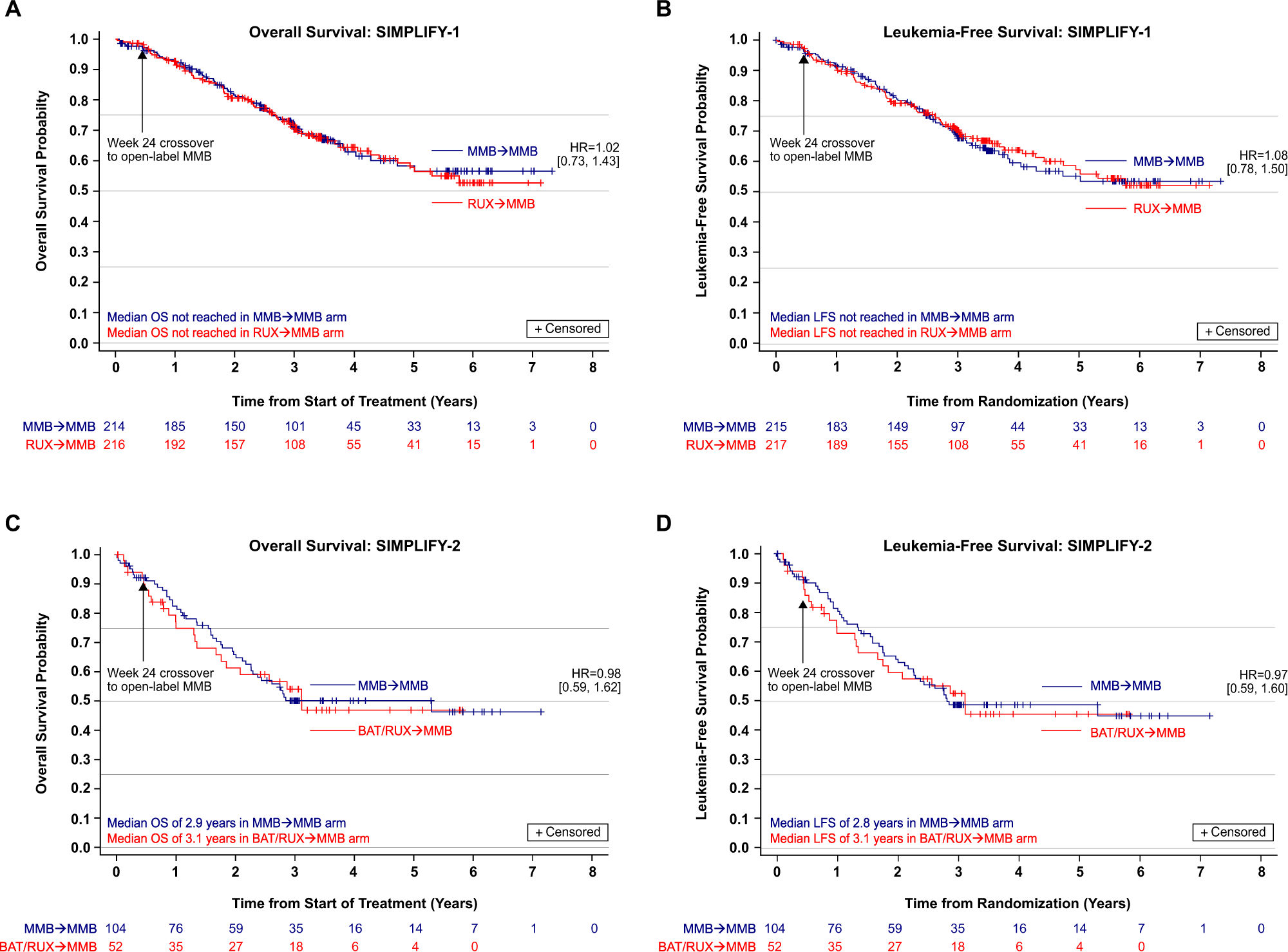
Overall survival in the SIMPLIFY-1 and SIMPLIFY-2 phase 3 trials of momelotinib in patients with myelofibrosis | Leukemia
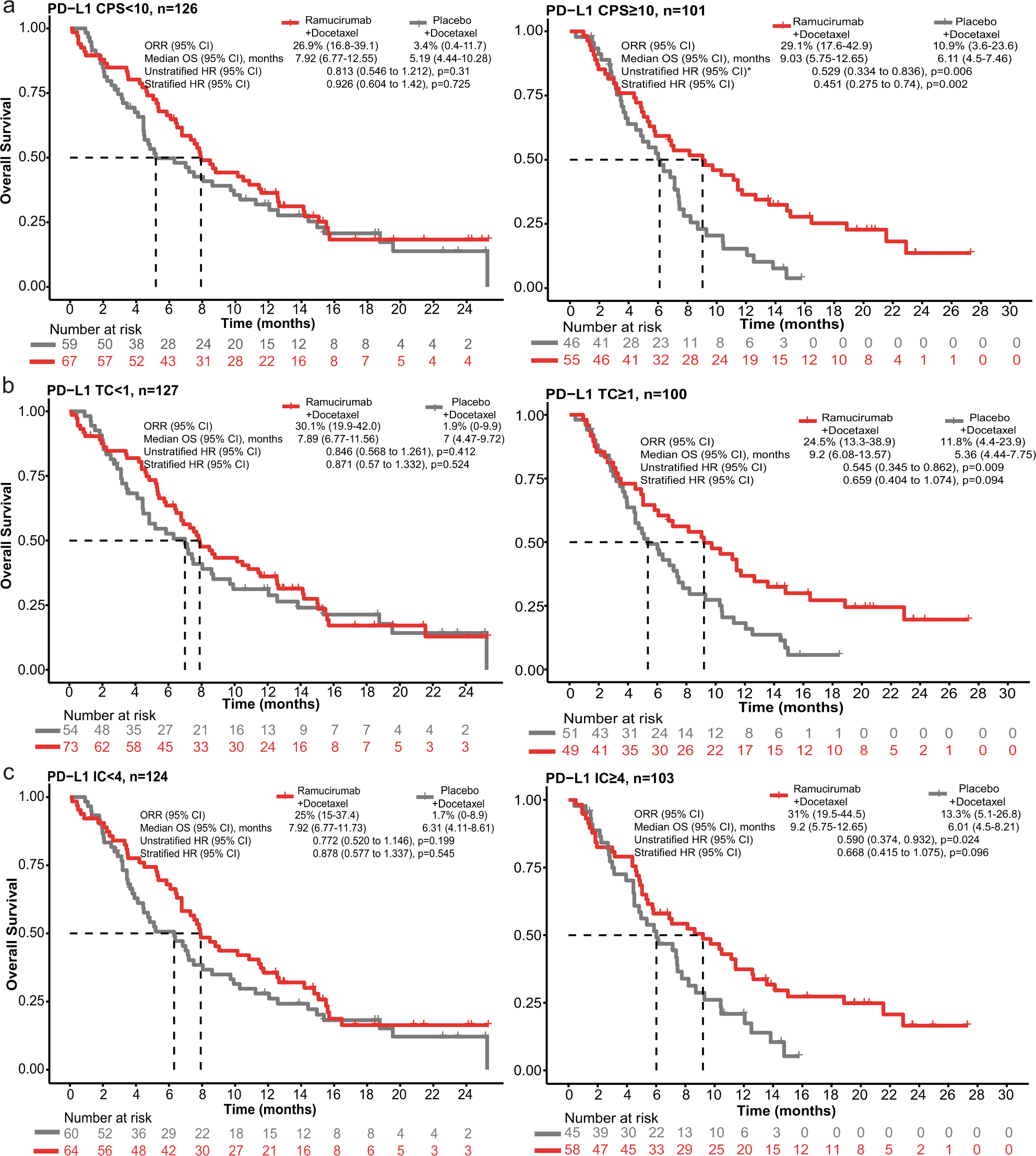
Predictive biomarkers for survival benefit with ramucirumab in urothelial cancer in the RANGE trial | Nature Communications
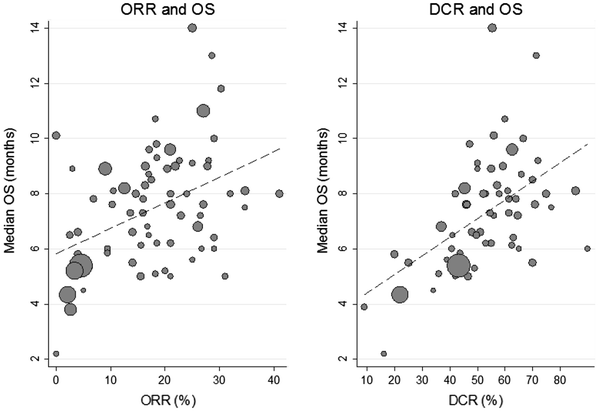
Correlation between overall survival and other endpoints in clinical trials of second-line chemotherapy for patients with advanced gastric cancer | SpringerLink
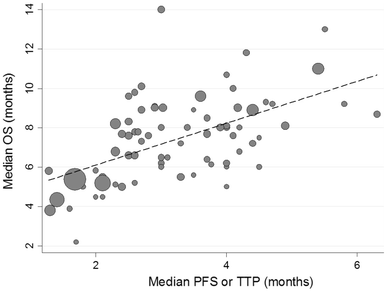
Correlation between overall survival and other endpoints in clinical trials of second-line chemotherapy for patients with advanced gastric cancer | SpringerLink