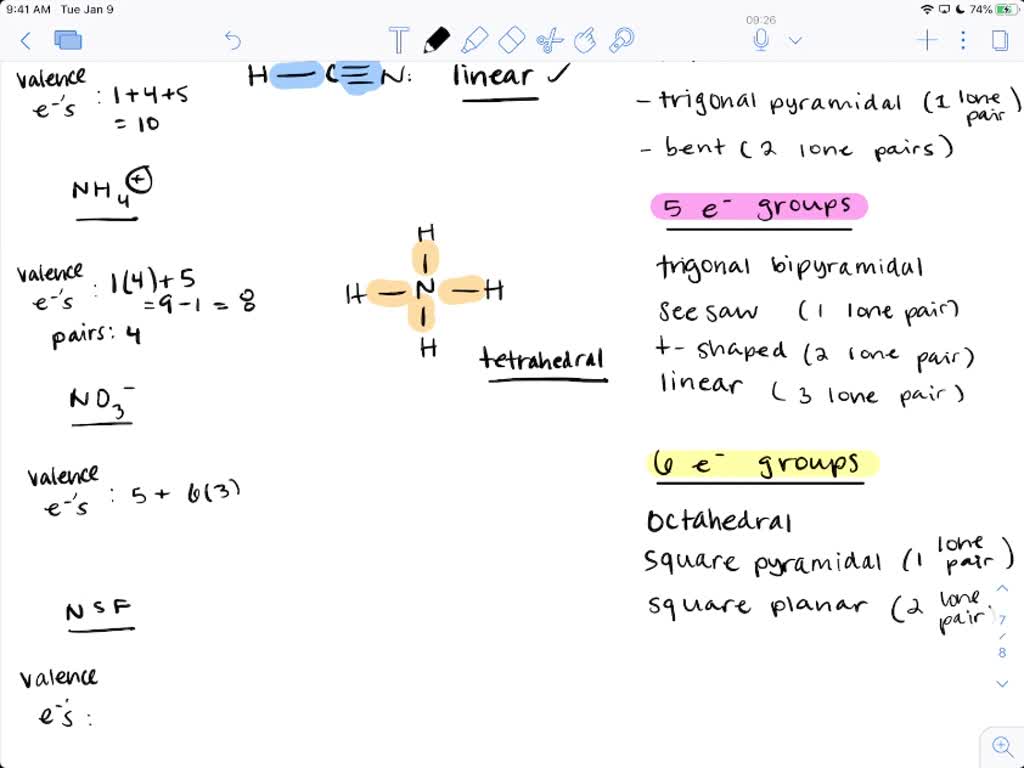
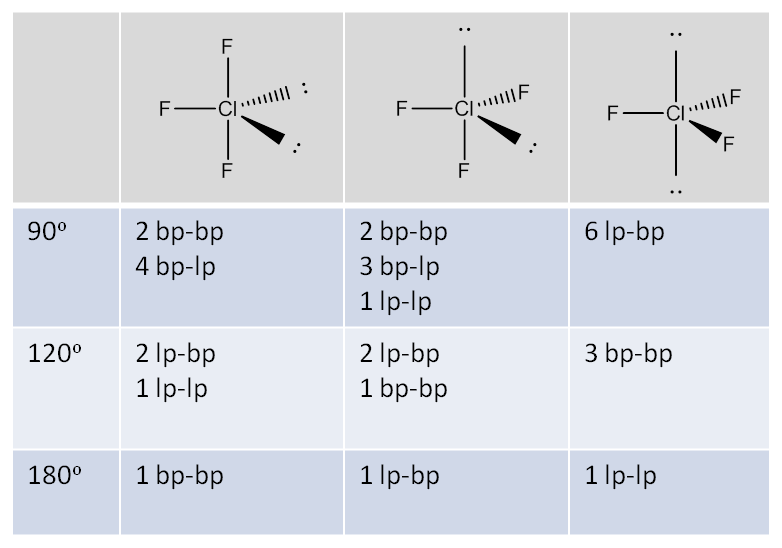
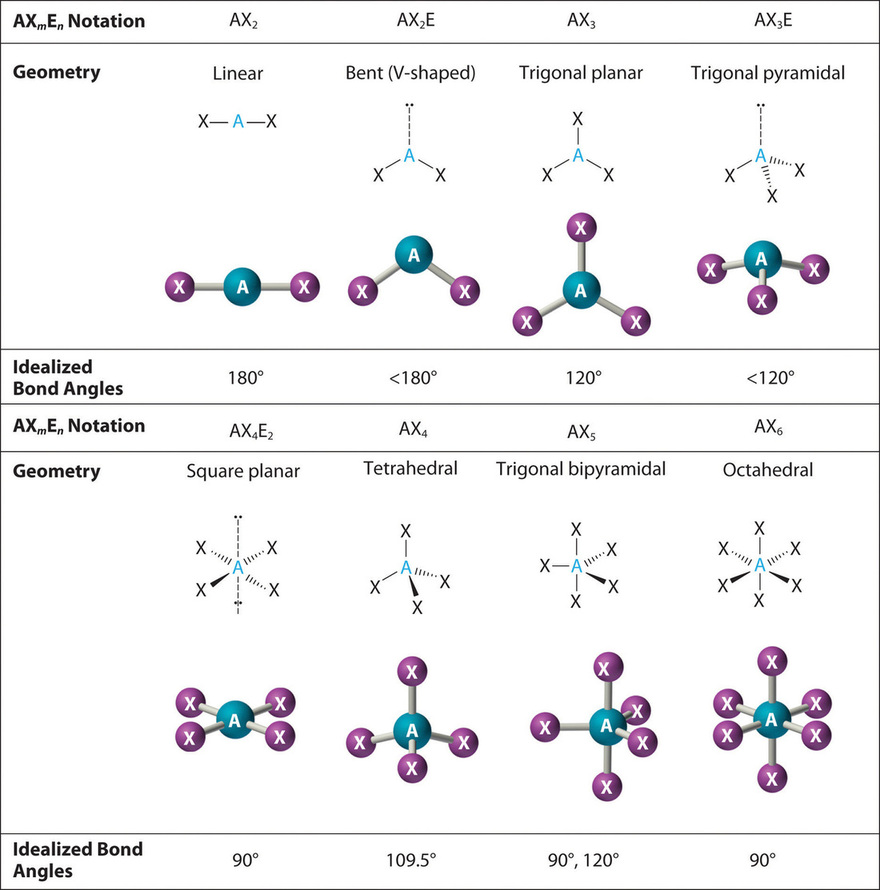
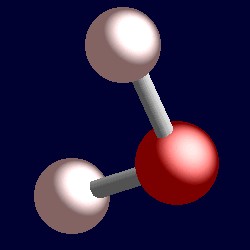
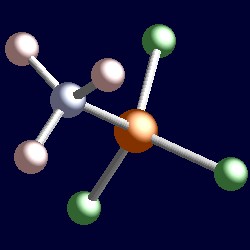
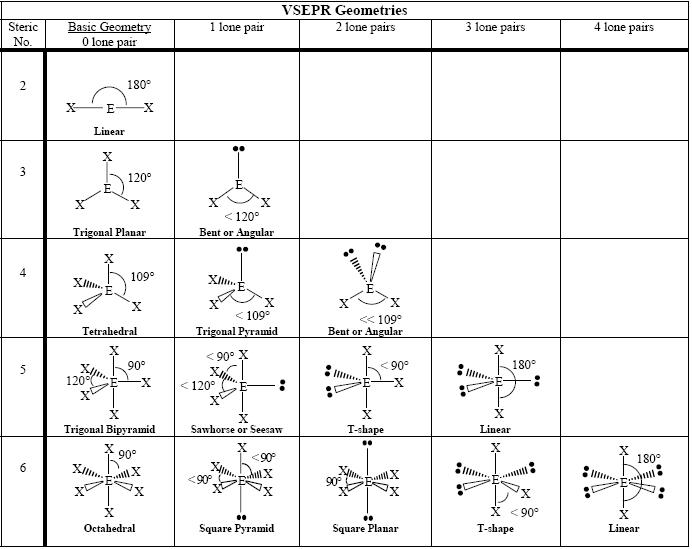
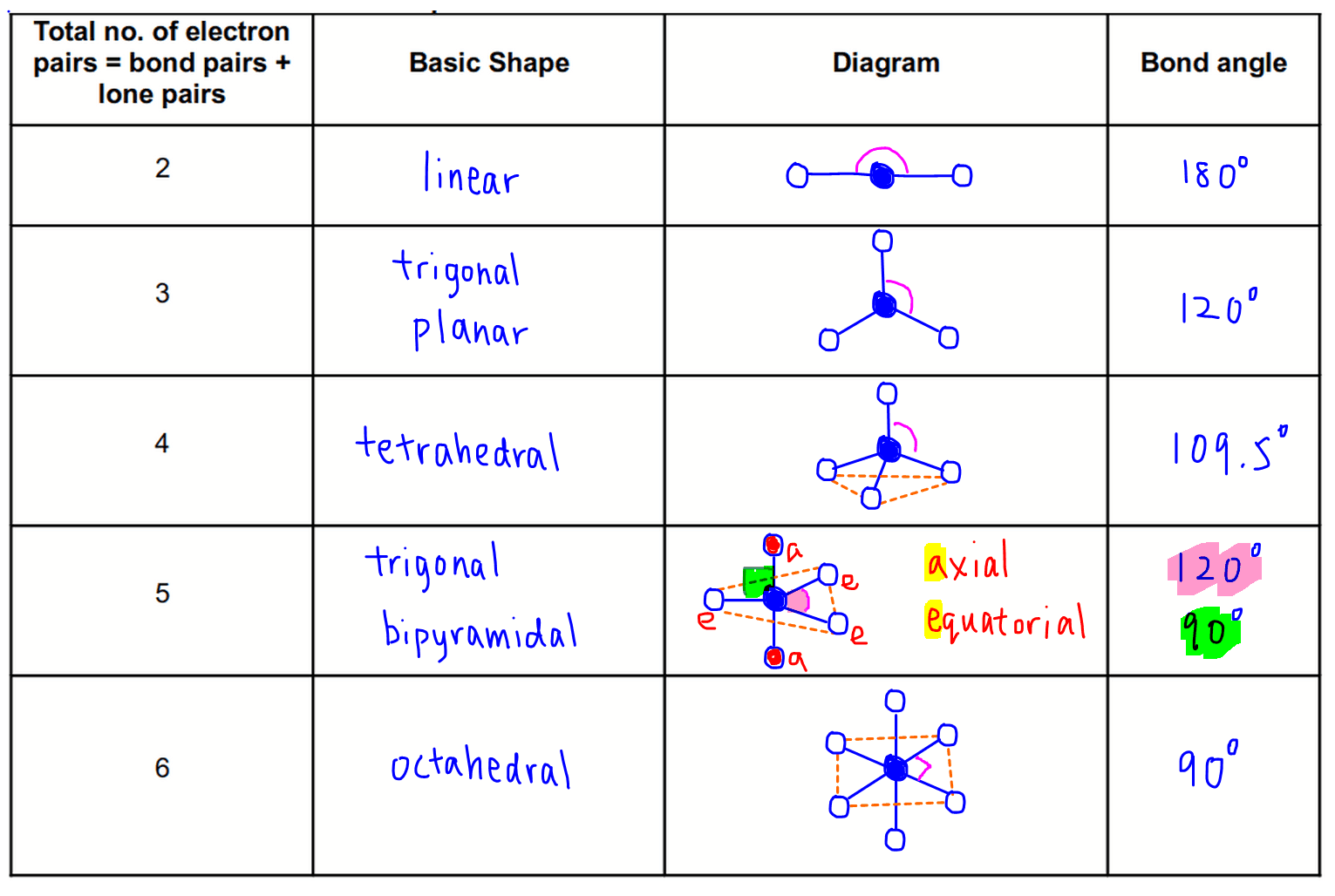
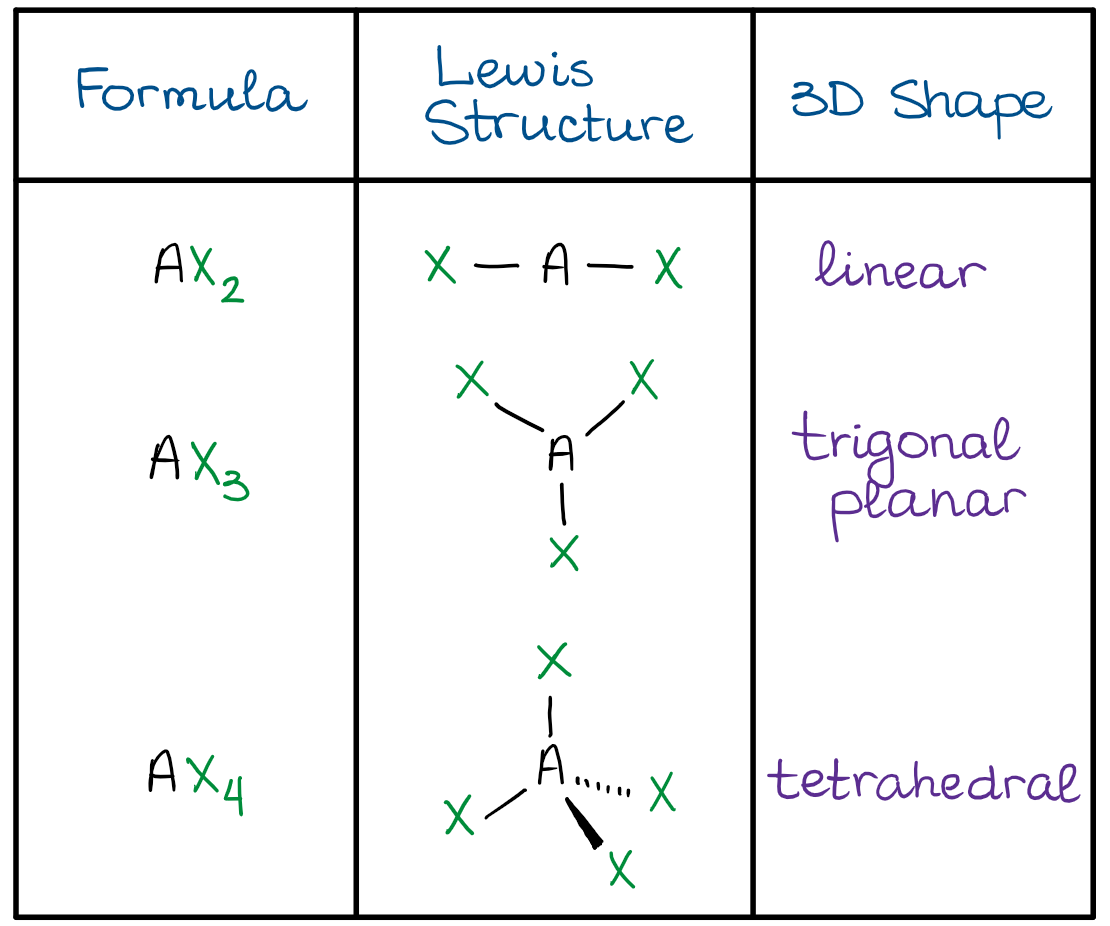
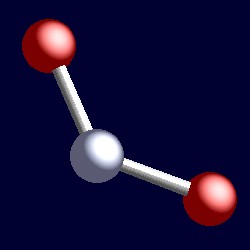
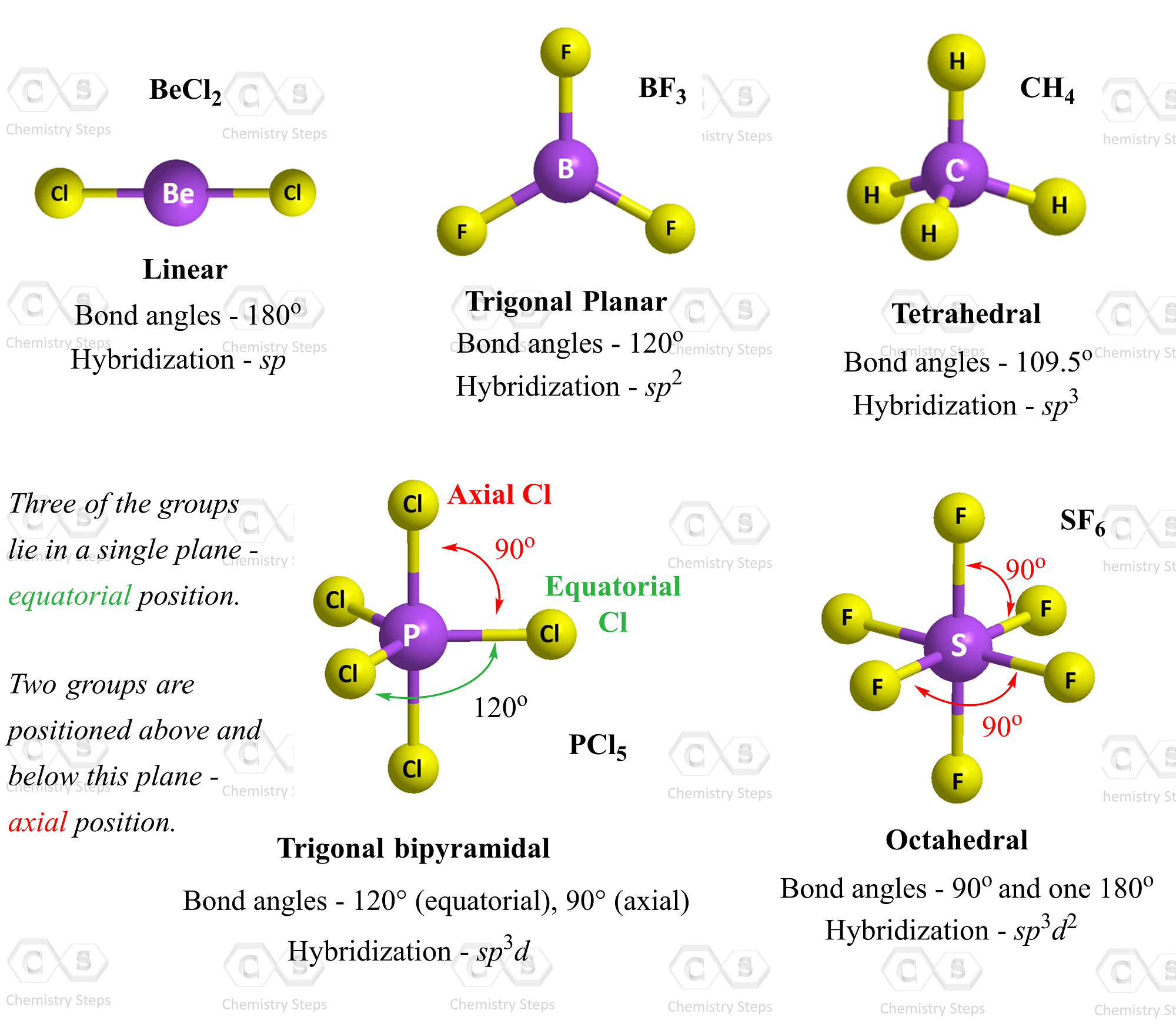
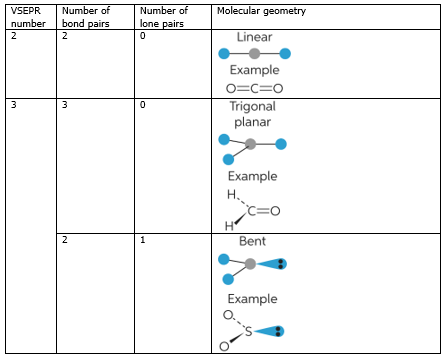
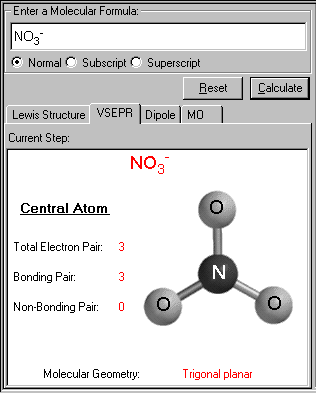
SOLVED: Name; Section: Experiment 7: VSEPR Structures, Molecular Modeling Lab assignment: Complete the first five rows ofthe tables below for 0z, Nz, CH+ HzO,BeClz, BFz; HzCO,COz, CIO4 , NOz , PCls, and
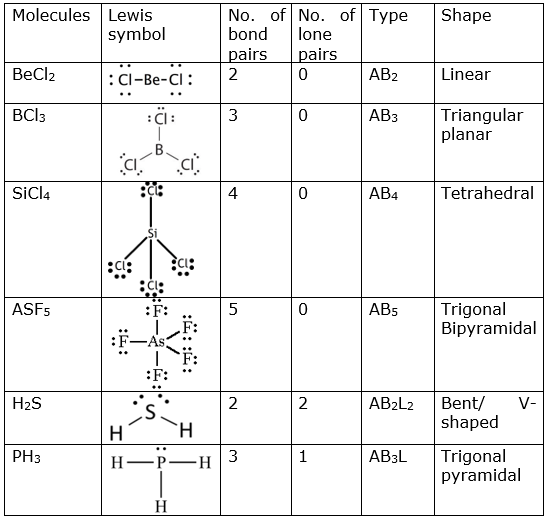
Discuss the shape of the following molecules using the VSEPR model: BeCl 2 , BCl 3 , SiCl 4 , AsF 5 , H 2 S, PH 3